Charlotte Martin, Luis E. Gimenez, Savannah Y. Williams, Yu Jing, Yiran Wu, Charlie Hollanders, Olivier Van der Poorten, Simon Gonzalez, Kevin Van holsbeeck, Santo Previti, Arthur Lamouroux, Suwen Zhao, Dirk Tourwé, Raymond C. Stevens*, Roger D. Cone*, and Steven Ballet*
J. Med. Chem. 2021, 64, 1, 357–369
Publication Date: November 14, 2020
https://doi.org/10.1021/acs.jmedchem.0c01620
Copyright © 2020 American Chemical Society
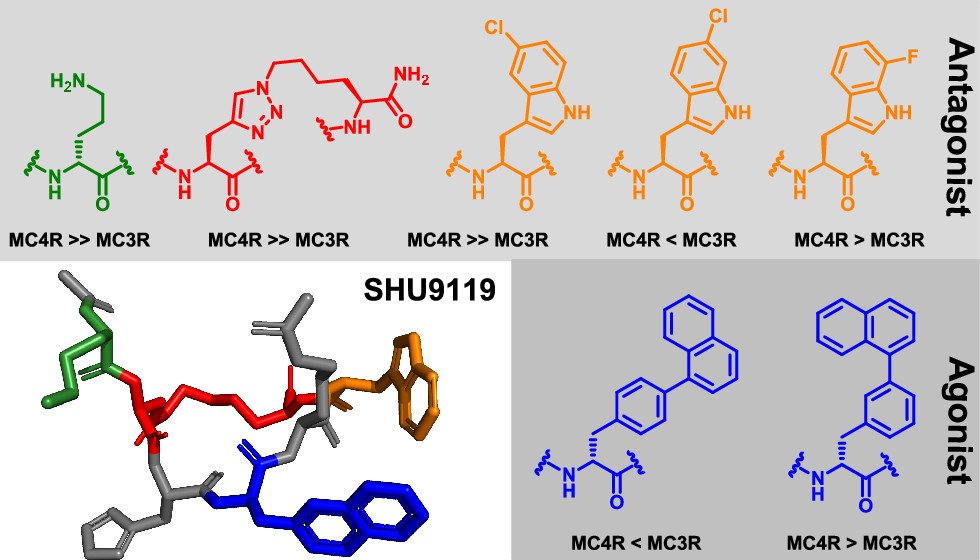
The melanocortin receptors (MC1R–MC5R) belong to class A G-protein-coupled receptors (GPCRs) and are known to have receptor-specific roles in normal and diseased states. Selectivity for MC4R is of particular interest due to its involvement in various metabolic disorders, including obesity, feeding regulation, and sexual dysfunctions. To further improve the potency and selectivity of MC4R (ant)agonist peptide ligands, we designed and synthesized a series of cyclic peptides based on the recent crystal structure of MC4R in complex with the well-characterized antagonist SHU-9119 (Ac-Nle4-c[Asp5-His6-DNal(2′)7-Arg8-Trp9-Lys10]-NH2). These analogues were pharmacologically characterized in vitro, giving key insights into exploiting binding site subpockets to deliver more selective ligands. More specifically, the side chains of the Nle4, DNal(2′)7, and Trp9 residues in SHU-9119, as well as the amide linkage between the Asp5 and Lys10 side chains, were found to represent structural features engaging a hMC4R/hMC3R selectivity switch.
Read the full paper in Journal of Medicinal Chemistry